The core difference between peptide and steroid hormones boils down to speed versus depth. Think of it this way: peptides are rapid, external messengers that trigger immediate cellular responses, while steroids act as deep, internal architects, directly altering genetic expression for sustained, long-term change. One is a quick command; the other is a core programming update.
The Core Difference: How Peptides and Steroids Work
When you compare peptide hormones vs steroid hormones, you're looking at two fundamentally different communication strategies in the body. It’s like sending a text message that demands an instant reply versus rewriting a company's entire mission statement. One creates a fast, temporary action; the other initiates a profound, lasting shift.
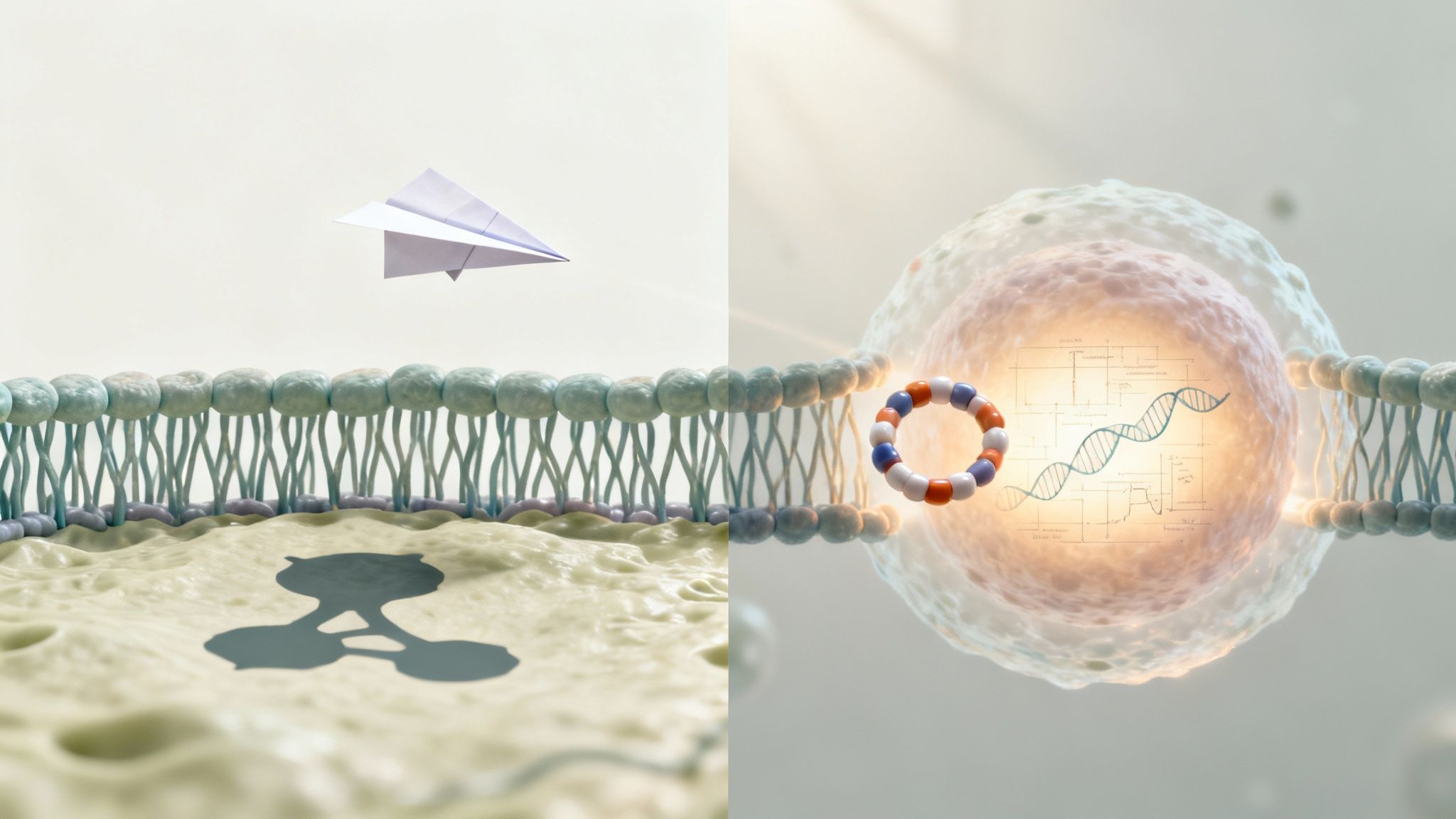
Peptide hormones are the body's rapid-response team. Since they're water-soluble, they can't simply pass through the fatty cell membrane. Instead, they deliver their message without ever entering the cell by binding to specific receptors on its surface, which then triggers a cascade of reactions inside.
Speed of Action
This surface-level interaction is precisely why peptide effects are felt so quickly. For researchers and biohackers, this molecular difference is critical. Peptides act like lightning-fast signals, whereas steroids are designed to play the long game by rewriting the very instructions in your DNA. By binding to cell surface receptors, peptides kick off rapid "second-messenger" cascades that can deliver noticeable effects in just seconds to minutes. You can find excellent primers on these mechanisms and their downstream effects for a deeper dive.
Depth of Impact
Steroid hormones operate with a completely different playbook. As derivatives of cholesterol, they are lipid-soluble, which gives them an all-access pass to glide right through the cell membrane. Once inside, they travel to the cell's nucleus, bind to intracellular receptors, and directly influence which genes are switched "on" or "off."
This genomic action is why steroid-induced changes, like muscle growth from testosterone, take hours or even days to initiate but are far more enduring. They aren’t just sending a signal; they are fundamentally altering the cell's protein-building instructions to force a long-term adaptation.
To put it plainly, peptides knock on the door and shout instructions. Steroids walk right inside, head to the main office, and rewrite company policy. Understanding this high-level distinction is essential before we dissect their specific structures, functions, and practical applications in research.
Peptide vs Steroid Hormones At a Glance
For a quick summary, this table breaks down the core characteristics of peptide and steroid hormones, giving you a high-level comparison at a glance.
| Characteristic | Peptide Hormones | Steroid Hormones |
|---|---|---|
| Origin | Amino Acid Chains | Cholesterol (Lipid) |
| Solubility | Water-Soluble (Hydrophilic) | Lipid-Soluble (Lipophilic) |
| Receptor Location | Cell Surface | Intracellular (Cytoplasm/Nucleus) |
| Mechanism | Second Messenger Cascade | Direct Gene Activation |
| Speed of Effect | Fast (Seconds to Minutes) | Slow (Hours to Days) |
| Duration of Effect | Short-Lived | Long-Lasting |
| Example | Insulin, Growth Hormone | Testosterone, Cortisol |
As you can see, their opposing traits in solubility, receptor location, and mechanism of action directly explain why they produce such different physiological outcomes. These foundational differences influence everything from administration routes to detection windows, which we'll explore next.
Understanding Their Chemical Blueprint and Origin
To really get a grip on the functional differences between peptide and steroid hormones, you have to start with their basic chemical makeup. A hormone's structure isn't just a technical detail; it’s the master plan that dictates where it comes from, how it travels through the body, and what it does when it gets to its target. One is built for speed in the watery highway of the bloodstream, while the other is designed to slip through fatty roadblocks and work from the inside.

This structural divide is the root cause of every other difference you'll see—from their mechanism of action to their half-life and how they're administered. For any researcher designing an experiment or just trying to make sense of the data, this molecular foundation is everything.
Peptide Hormones: The Water-Soluble Messengers
At their core, peptide hormones are just short chains of amino acids, the very same building blocks that make up proteins. You can think of them as tiny, highly specialized protein messengers, ranging from just a handful of amino acids to a few dozen. This amino-acid base makes them hydrophilic, meaning they love water.
This single characteristic is a game-changer. Our bloodstream is mostly water, so peptide hormones can dissolve right in and travel freely. No need for a special transport shuttle. This allows them to get from their point of release to their target tissue incredibly quickly.
But this water-solubility has one major drawback. Cell membranes are made of lipids (fats), which create an oily barrier that pushes water-loving molecules away. Because of this, peptide hormones cannot pass through the cell membrane on their own.
Steroid Hormones: The Fat-Soluble Infiltrators
Steroid hormones couldn't be more different. They are all derived from a single parent molecule: cholesterol. Their foundational structure is a lipid, which makes them lipophilic, or fat-soluble. This simple fact puts them in a completely different league with a unique set of rules.
Being fat-soluble means they are hydrophobic—they don't mix well with water. This creates a problem for traveling through the bloodstream. To get around this, they have to latch onto carrier proteins, like albumin, which act as their personal chauffeurs, escorting them safely to their destinations.
The real power of being lipophilic comes into play at the target cell. A steroid hormone's fatty nature allows it to glide right through the lipid-based cell membrane, giving it a direct backstage pass to the cell’s internal machinery.
This fundamental difference in their relationship with water and fat is the primary driver behind their completely separate modes of action.
Structural Comparison At a Glance
The molecular distinction between these two classes of hormones dictates their entire lifecycle. Here’s a quick breakdown of their core structural differences and what it means for how they function:
Peptide Hormones:
- Building Block: Chains of amino acids.
- Solubility: Water-soluble (hydrophilic), letting them dissolve and travel easily in the blood.
- Cell Interaction: Can't cross the fatty cell membrane; they have to bind to receptors on the cell's surface.
- Analogy: A key that fits a specific lock on the outside of a door.
Steroid Hormones:
- Building Block: Derived from cholesterol (a lipid).
- Solubility: Fat-soluble (lipophilic), which means they need carrier proteins to move through the blood.
- Cell Interaction: Can easily slip through the cell membrane to act on receptors inside the cell.
- Analogy: A master key that can pass right through the door to get to the main control room.
Ultimately, their chemical blueprint is their destiny. The water-loving nature of peptides makes them perfect for fast, external signaling. In contrast, the fat-loving nature of steroids allows them to become deep, internal regulators of a cell's genetic programming. This is the central concept you need to grasp to understand why they are researched and applied so differently in performance, medicine, and recovery.
How They Talk to Your Cells
Beyond their chemical makeup, the real difference between peptide and steroid hormones is how they communicate with your cells. It's like comparing a text message to a software update. One is a quick command shouted from outside the cell, while the other is a slow, deliberate rewrite of the cell's fundamental programming.

This isn't just a trivial detail. It’s the core reason peptides are masters of rapid, acute effects, while steroids are the go-to for deep, long-lasting transformations. If you want to predict what a compound will do, you have to understand its signaling pathway.
Peptides: The Messengers at the Gate
Because peptide hormones are made of amino acids, they're water-soluble. This means they can't just waltz through the fatty, lipid-based membrane of a cell. They’re stuck outside.
So, how do they get their message across? By knocking on the door. They bind to specific receptor proteins sitting on the cell's surface.
Think of it like a key turning in a lock on a door. The key doesn't go inside the house, but it triggers an alarm system—a cascade of signals—within. Once the peptide binds, the receptor changes shape and kicks off a chain reaction inside the cell using what are called second messengers (like cyclic AMP). This process amplifies the original signal and triggers immediate changes, like switching enzymes on or off.
This is a non-genomic pathway, meaning the hormone never even enters the cell’s nucleus or messes with DNA. It's all about speed and efficiency, perfect for things that need to happen now, like managing blood sugar or triggering a sudden hormone release.
This is why the effects of peptides like Ipamorelin are felt so quickly. They operate through this rapid-fire signaling cascade. For a practical look at what this means in a research setting, you can check out our guide covering Ipamorelin before and after observations.
Steroids: The Architects Inside the Cell
Steroids play a completely different game. Their fatty, lipid-soluble structure gives them an all-access pass right through the cell membrane. No need to knock.
Once inside, they travel through the cell’s interior to find their target: intracellular receptors. When a steroid molecule links up with its receptor, they form a powerful complex that marches straight into the nucleus—the cell's command center.
Here's where the magic happens. This hormone-receptor complex binds directly to DNA at specific sites called Hormone Response Elements (HREs). In doing so, it acts like a genetic switch, directly influencing gene transcription. It tells the cell which proteins to make and which to stop making. This is a fundamental reprogramming of the cell's function and structure from the inside out.
Building new proteins takes time, which is why the effects of steroids are much slower to appear but are far more profound and lasting.
Comparing Their Signaling Mechanisms
The contrast in their communication styles couldn't be starker, and it dictates everything about how they are used in research and performance. One is for the here and now; the other is for foundational change.
Peptide Hormone Signaling (The "Text Message")
- Action Type: Non-genomic. It doesn't touch the DNA.
- Receptor Location: On the cell's outer surface.
- Mechanism: Activates second messengers for rapid, enzyme-level changes.
- Timeframe: Incredibly fast. We're talking seconds to minutes.
- Outcome: Acute, short-lived adjustments to cell function.
Steroid Hormone Signaling (The "Software Update")
- Action Type: Genomic. Directly interacts with DNA to change gene expression.
- Receptor Location: Inside the cell (cytoplasm or nucleus).
- Mechanism: Forms a complex that acts as a transcription factor, controlling protein synthesis.
- Timeframe: Much slower. It can take hours or even days to see the full effects.
- Outcome: Deep, long-lasting structural and functional changes.
Ultimately, these two paths explain their distinct roles in the body. Peptides are the tactical first responders, handling immediate threats and opportunities. Steroids are the strategic planners, initiating deep-seated changes that redefine what a cell is and what it can do for the long haul.
4. Practical Insights: Administration and Half-Life
Theory is one thing, but what happens when the rubber meets the road? For any researcher, the real-world application is what truly matters. How you handle and administer these compounds, how long they stick around in a system, and how often you need to dose them—these aren't just minor details. They are the critical variables that will make or break your experimental protocol.
The core of it all comes down to solubility and stability. Peptides are essentially delicate, water-soluble protein chains. Steroids are rugged, fat-soluble molecules built from cholesterol. This fundamental difference creates two entirely different worlds when it comes to practical use.
The Fleeting Nature of Peptide Hormones
Peptide hormones are the sprinters of the endocrine world. They have an incredibly short half-life, often lasting just a matter of minutes. Think of something like a Growth Hormone Releasing Hormone (GHRH) analogue—its activity might peak and fade in less than 30 minutes. Others, like Growth Hormone itself, might hang around for a few hours.
This short-lived presence is a classic double-edged sword. On one hand, it allows for incredible precision. You can create sharp, controlled pulses that mimic the body's natural rhythms. But on the other hand, it means you're constantly chasing a moving target. To maintain stable, effective levels, a researcher might need to administer a peptide multiple times a day.
Then there's the issue of their fragile structure. Being proteins, they're sitting ducks for your digestive enzymes.
Swallow a peptide, and your stomach acid and enzymes will tear it apart into basic amino acids before it ever has a chance to work. It becomes expensive, useless protein powder. That's precisely why peptides have to be injected, usually subcutaneously, to slip past the digestive system and get right into the bloodstream.
This delivery method ensures the molecule actually reaches its target receptors intact. It also means they demand careful handling—refrigeration and proper reconstitution are non-negotiable to preserve their delicate structure. If you need a refresher, our guide on how to reconstitute peptides correctly walks you through the process step-by-step.
The Enduring Presence of Steroid Hormones
Steroids, in contrast, are built for the long haul. Because they’re derived from the highly stable cholesterol molecule, they are much more resistant to being broken down by the body. Their half-life isn't measured in minutes, but in hours or even days, especially when an ester is involved. Attaching an ester is a common chemical trick that essentially creates a time-release mechanism, slowing the hormone's absorption from the injection site.
This durability completely changes the game from a practical standpoint:
- Less Frequent Dosing: An esterified steroid like Testosterone Enanthate might only need to be administered once or twice a week to keep blood levels rock-solid.
- Different Routes: Their oily nature is perfect for deep intramuscular injections, where they can form a small depot that releases the compound slowly and steadily over time.
- Oral Viability (With a Big Catch): Some steroids can be modified to survive the "first pass" through the liver, making them orally active. This modification (called 17-alpha-alkylation) is effective, but it comes at a steep price: a significant increase in liver toxicity. This is a massive safety consideration for any research model.
When it comes down to peptide hormones vs steroid hormones, the day-to-day practicalities of administration and half-life are often the deciding factor. Peptides give you surgical precision and rapid, targeted effects, but they demand a high-maintenance schedule and meticulous handling. Steroids deliver powerful, sustained action with far less fuss, but their long-lasting nature can bring a greater risk of systemic side effects and widespread hormonal shifts.
Ultimately, your research objective dictates the right tool for the job. Are you studying an acute, short-term signaling pathway, or are you investigating long-term changes in physiology? The answer will point you to the right pharmacokinetic profile.
Comparing Their Impact on Performance and Recovery
When you move past the biochemistry and into the real world, the differences between peptide and steroid hormones aren't just academic—they're dramatic. It’s like comparing a scalpel to a sledgehammer. Both are powerful tools, but you'd never use one where the other is needed. Their effects on performance, recovery, and the body as a whole couldn't be more distinct.
For researchers focused on performance, peptides offer a level of precision that's simply not possible with traditional anabolics. They're designed to hit a specific receptor or trigger a single pathway, delivering a clean, targeted outcome with minimal collateral damage. That specificity is their defining feature.
Steroid hormones, on the other hand, bring overwhelming force. By directly manipulating gene expression, they unleash widespread anabolic and androgenic changes across the entire system. The result is a powerful, undeniable effect, but it’s far from precise.
Peptides: The Targeted Specialists
Peptides shine when the goal is a nuanced, specific physiological change. Their specialization makes them perfect for research into everything from accelerated tissue repair to fine-tuning metabolic function.
Here are a few classic examples from the lab:
- Targeted Tissue Repair: A research peptide like BPC-157 is a perfect illustration. It’s studied for its incredible ability to speed up healing in specific tissues—tendons, ligaments, muscles—without causing broad, systemic muscle growth. It goes right to the site of injury and gets to work.
- Growth Hormone Pulsing: Secretagogues like Ipamorelin or CJC-1295 don't flood the system with synthetic growth hormone. Instead, they’re designed to prompt the pituitary gland into releasing a natural pulse of GH, mimicking the body's own rhythm to support recovery, enhance sleep, and aid fat loss.
- Metabolic Regulation: Peptides like the GLP-1 agonists are researched for their profound effects on blood sugar and appetite. They target very specific receptors in the brain and gut, offering a focused way to investigate metabolic health.
This level of precision is the key advantage. A researcher can isolate and study a single biological process—like collagen synthesis or fat oxidation—without setting off the body-wide hormonal cascade that comes with steroids. If you want to dive deeper into how specific peptides influence muscle development, our guide on the best peptides for muscle growth breaks it all down.
Steroids: The Systemic Powerhouses
Steroid hormones, especially anabolic-androgenic steroids (AAS), are the heavy artillery. Their primary benefit in performance research comes from their unmatched ability to crank up muscle protein synthesis across the board.
When a molecule like testosterone gets into a muscle cell, it essentially rewrites the instructions, telling the cell's DNA to build more protein. This leads to massive gains in muscle size (hypertrophy) and raw strength. This isn't a localized effect; it happens in nearly every skeletal muscle, resulting in a profound and systemic transformation.
Of course, that systemic power has systemic consequences. The same androgenic signals that build muscle also affect skin, hair follicles, and the prostate, among other things. This powerful, all-encompassing action is the fundamental trade-off when you're comparing peptides and steroids.
For a researcher, the choice often boils down to a single question: Are you trying to fine-tune a specific biological process with minimal side effects, or are you aiming to trigger a powerful, body-wide anabolic state?
This decision path helps visualize that choice. It shows when to consider one class of compounds over the other based on whether the research goal is a quick, targeted effect or a long-lasting, systemic one.
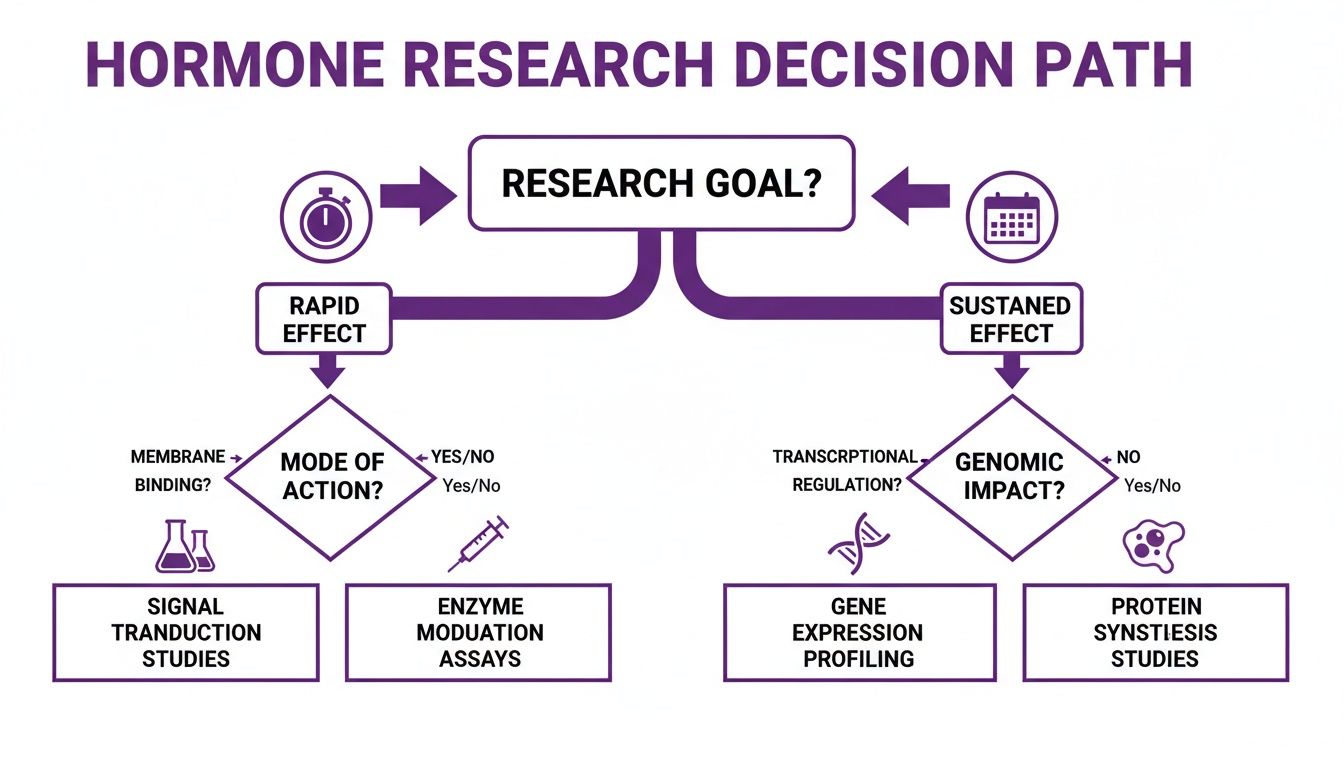
As the chart makes clear, peptides are the logical choice for acute, precision-focused studies. Steroids, however, are better suited for research into profound, long-term physiological changes.
Comparative Effects in a Performance Research Context
To really get a feel for their distinct roles, nothing beats a side-by-side comparison. The table below lays out how the different mechanisms of peptides and steroids translate into practical outcomes in a research setting.
| Effect Category | Peptide Examples (e.g., Ipamorelin, BPC-157) | Steroid Examples (e.g., Testosterone) |
|---|---|---|
| Muscle Growth | Indirect or supportive; can aid recovery and GH release, but rarely drives primary hypertrophy. | Direct and powerful; significantly increases muscle protein synthesis for systemic hypertrophy. |
| Strength Gains | Minimal direct impact on raw strength; effects are secondary to improved recovery or joint health. | Primary effect; directly enhances neuromuscular efficiency and muscle fiber size, leading to major strength increases. |
| Tissue Repair | Highly targeted and potent; peptides like BPC-157 are studied specifically for accelerated healing. | Indirect support through general anabolism, but lacks the specialized, localized repair action of peptides. |
| Fat Loss | Can be highly effective through specific pathways (e.g., stimulating GH release or improving insulin sensitivity). | Can aid in fat loss by increasing metabolic rate and muscle mass, but is not its primary function. |
| Systemic Impact | Low systemic impact; actions are generally confined to the targeted pathway. | High systemic impact; affects multiple organ systems due to its broad androgenic and anabolic signaling. |
Ultimately, this comparison highlights the core difference: peptides offer targeted solutions for specific problems, while steroids provide a broad, systemic push for overall mass and strength. Each has a clear and distinct role in a research context.
Navigating Safety Profiles and Potential Side Effects
In serious research, understanding what can go wrong is just as critical as knowing what could go right. When you put peptides and steroids side-by-side, their safety profiles and potential side effects are worlds apart—a direct reflection of their completely different mechanisms. Any researcher worth their salt needs to have a firm grip on these differences to design experiments that are not just effective, but also ethical and predictable.
The core of the matter really boils down to localized versus systemic effects. Many peptides are known for a more targeted, predictable, and often milder range of side effects. Steroids, on the other hand, have a well-earned reputation for causing widespread, systemic issues that can impact multiple organ systems all at once.
This isn't by accident. It's a direct result of how they work. Peptides knock on the cell's door by binding to surface receptors, limiting their influence. Steroids barge right in, get into the nucleus, and start rewriting gene expression, setting off a cascade of effects throughout the entire body.
The More Confined Risks of Peptides
When it comes to research peptides, the most common adverse effects tend to be localized and short-lived. Often, they're tied directly to the act of administration itself.
Here's what to look out for with peptides:
- Injection Site Reactions: A bit of redness, swelling, or irritation right where you inject is common. It’s a local immune response and typically clears up on its own pretty fast.
- Transient Effects: Some peptides, especially the growth hormone secretagogues, might cause some temporary water retention, a feeling of pins and needles (paresthesia), or a brief spike in cortisol or prolactin. These are almost always dose-dependent and fade as the body gets used to the compound.
- Purity and Contamination: Let's be blunt: the biggest danger in peptide research often isn't the peptide itself, but what's mixed in with it. Shady sourcing can introduce contaminants that lead to completely unpredictable and dangerous outcomes. Sourcing high-purity, third-party tested materials is non-negotiable if you want reliable and safe data.
The Systemic Risks of Steroid Hormones
Steroid hormones, particularly the anabolic-androgenic steroid (AAS) class, bring a much longer and more serious list of potential problems to the table. Their power is systemic, and so are their risks.
The elephant in the room with anabolic steroid research is the suppression or complete shutdown of the Hypothalamic-Pituitary-Testicular Axis (HPTA). When you introduce outside androgens, the body's natural testosterone factory shuts down. This can be a long-term problem that persists well after the compound is stopped, often requiring complex protocols to restore function.
Beyond HPTA shutdown, there are other major risks to consider:
- Cardiovascular Strain: Steroids have been shown time and again to wreck cholesterol profiles. They drive up LDL ("bad" cholesterol) while tanking HDL ("good" cholesterol), which is a recipe for long-term cardiovascular trouble.
- Hepatotoxicity (Liver Damage): This is a huge concern with oral steroids, specifically the 17-alpha-alkylated versions. They’re engineered to survive the first pass through the liver, but that resistance comes at the cost of putting immense strain on the organ.
- Androgenic Side Effects: These are the classic signs of high androgen levels: acne, accelerated male pattern baldness in those genetically prone, and potential prostate enlargement.
At the end of the day, comparing the safety of peptides and steroids means accepting two very different categories of risk. Peptides typically present localized, more manageable issues. Steroids, however, carry profound systemic risks that demand a deep understanding and a cautious approach in any research context.
Common Questions About Peptides and Steroids
As we dive deeper into hormonal signaling, the practical questions always start to surface. The whole peptide vs. steroid debate isn't just a textbook exercise; it has real-world consequences for how research is designed and what the results actually mean. This section cuts through the noise to tackle the most common questions I hear.
Let's clear the air on a few things. Understanding the fundamental differences between these molecules is key, as it impacts everything from their safety profiles to how they can be properly used in a controlled lab setting.
Are Peptides Really a Safer Alternative to Steroids for Research?
Generally speaking, yes, peptides are often seen as having a better safety profile in a research environment. The main reason is their specificity. Peptides tend to act on very narrow targets, so you don't get the same kind of systemic hormonal fallout you see with anabolic steroids. A good example is BPC-157, which hones in on tissue repair pathways without throwing the entire endocrine system out of whack.
Anabolic steroids, on the other hand, can completely shut down the body's own hormone production, which creates a whole host of complexities for clinical studies, including the need for careful post-protocol management. That said, let's be clear: all compounds have potential side effects. Safety is never a given. It always comes down to the purity of the compound, the precision of the dosage, and responsible lab practices. Remember, all products discussed here are strictly for research use only.
Can You Study Peptides and Steroids Together?
Absolutely. In more advanced research models, investigating how peptides and steroids work together is a fascinating and common field of study. A researcher might, for instance, look at how a peptide that stimulates growth hormone release could work synergistically with the powerful muscle-building effects of an androgenic steroid.
This kind of research opens the door to studying complex biological interactions. You might see a peptide like BPC-157 used to see if it can support joint and tendon integrity under the simulated stress that would be associated with anabolic research. These are sophisticated experimental designs that demand a thorough understanding of both classes of compounds.
Why Do Peptides Need to Be Injected, but Some Steroids Can Be Taken Orally?
This all comes down to their basic chemistry and how fragile they are. Peptides are essentially short chains of amino acids—think of them as small, delicate proteins. If you were to swallow them, the powerful digestive enzymes in your stomach would tear them apart into useless bits before they ever had a chance to enter the bloodstream. Injection is simply a way to bypass that destructive digestive process.
Steroids are a different beast entirely. Being lipid-based, they are much tougher. While many are still injected to get the best bioavailability, some can be chemically tweaked (a process known as 17-alpha-alkylation) to survive the first pass through the liver. This modification is what makes oral administration possible, but it comes at a cost—it often dramatically increases the potential for liver toxicity, a critical factor to account for in any research protocol.
For researchers who demand high-purity, third-party verified compounds for their work, Bullit Peptides offers a comprehensive catalog of research-grade peptides built for rigorous scientific investigation. You can explore our selection and find detailed product information on our website.





