When a company claims its new peptide builds muscle or a supplement cranks up your metabolism, how do they really know? The truth is, without hard data, it’s all just marketing. The answer—and the gold standard for proof—lies in a powerful technique called labeled protein synthesis.
It’s a bit like putting a microscopic tracking device on the individual building blocks of protein, the amino acids. By "tagging" these raw materials, scientists can watch in real-time as your body uses them to build new tissue.
This guide pulls back the curtain on how researchers track this fundamental biological process. You'll understand the science that separates groundbreaking compounds from wishful thinking, providing definitive proof for everything from post-workout muscle repair to the metabolic shifts driven by cutting-edge peptides. This is where biology's blueprint delivers real-world results.

Inside The Cellular Factory
At its core, labeled protein synthesis is a method for measuring one of the most vital activities in the body. Inside every cell, tiny molecular machines called ribosomes work tirelessly, assembling proteins by linking amino acids together according to instructions from our genes. Think of them as cellular factories.
The big question for scientists has always been: how do we measure the output of these factories? How can we tell the difference between a brand-new protein and an old one that's been around for a while? That's precisely where labeling comes into play.
By introducing specially "tagged" amino acids into a system—whether it’s cells in a petri dish or a human volunteer—we can see exactly which proteins are new. If the finished proteins contain our tag, it’s undeniable proof they were just built.
Why This Is a Game-Changer for Performance Science
This isn't just some obscure lab technique; it's the definitive method for validating any compound that claims to influence growth, metabolism, or repair. It gives us clear, unambiguous answers to the most critical questions in performance and medicine.
- Muscle Growth: Does that new peptide actually trigger an increase in muscle protein synthesis after a workout? Labeling gives you a number, not a guess.
- Metabolic Health: How exactly do drugs like GLP-1 agonists change body composition? Labeling shows us how they affect protein turnover in muscle, fat, and other organs.
- Drug Discovery: Can a new therapeutic stimulate the production of vital proteins to combat a disease? This is how we find out.
By providing a clean, quantifiable number—the rate at which tagged amino acids become new proteins—this technique moves us beyond simple correlation and into the realm of causation. It’s the difference between thinking a compound works and proving it. This is the hard data that powers all legitimate modern research.
The Scientific Roots Of Tracking Protein Creation
Every modern method for optimizing human performance—from targeted peptides to precise nutritional timing—is built on a foundation laid by true scientific pioneers. The ability to watch a protein being built in real-time wasn't just invented in some sleek, modern lab. It was born from gritty, hands-on experiments decades ago, and understanding this journey into labeled protein synthesis shows the raw ingenuity that first unlocked our cellular secrets.
Picture the scientific world just after World War II. The cell's inner workings were still a massive black box. Researchers knew proteins were vital, but how they were made was a complete mystery. This is where people like Paul Zamecnik came in, not with supercomputers, but with a brand-new tool that would change everything: radioactivity.
The Dawn Of Radioactive Tracing
In the late 1940s and early 1950s, Zamecnik and his colleagues sparked a revolution. They started using radiolabeled amino acids to follow their path inside a cell. Before you could just order these tracers from a catalog in the late 1950s, these scientists had to synthesize them from scratch, using basic chemicals like formaldehyde. It was that kind of old-school grit that led to one of the biggest leaps in biology.
By using radioactive isotopes like carbon-14, these teams could "tag" an amino acid and literally watch it get woven into a new protein. For the very first time, they could tell a brand-new protein apart from all the others, giving them a direct window into the cell’s assembly line.
In 1951, Zamecnik’s lab at Massachusetts General Hospital created the first-ever cell-free system from rat liver tissue. This was a game-changer, allowing them to study protein synthesis without the chaos of a living cell. They quickly found that the process needed energy; synthesis rates shot up when they added ATP and GTP. You can dive into the original research to see how these foundational experiments were set up on the National Library of Medicine website.
Pinpointing The Protein Factory
With a system that worked, the next logical question was where was all this happening? The team meticulously broke down the cell's components and tested each fraction one by one. By 1954, they had their answer. The "microsomal fraction," which we now recognize as the ribosomes on the endoplasmic reticulum, was the undisputed hub of protein creation.
This discovery was monumental. They hadn't just proven you could measure protein synthesis; they had found the factory floor. The ribosome was officially crowned the site of this incredible biological process.
- The Blueprint: These experiments confirmed amino acids were the direct building blocks for proteins.
- The Fuel: They showed the process was energy-dependent, needing ATP and GTP to run.
- The Location: They identified the ribosome as the specific cellular machine doing the work.
The final piece of this initial puzzle was determining speed. Using a brilliant method called a pulse-chase experiment, they introduced a short "pulse" of radioactive amino acids, then "chased" it with a flood of normal, non-radioactive ones. By tracking the radioactive signal, they could clock the whole operation. The results were stunning: in bacteria, an entire protein could be built in just 5 to 10 seconds.
These early discoveries didn't just kickstart a new field. They gave us the blueprint for the engine of life itself. Every insight we have today—about muscle growth, metabolic health, and how drugs work—stands on the groundwork laid by these pioneers and their ingenious use of labeled protein synthesis.
Decoding The Blueprint For Protein Synthesis
Early on, scientists figured out that proteins were being built and even where they were made inside the cell. But the million-dollar question remained: how did the cell know which specific protein to assemble? The instructions were clearly locked away in our DNA, but it was a four-letter code that somehow had to direct the precise arrangement of over 20 different amino acids.
Imagine finding an encrypted message (the DNA) and the final, translated text (the protein), but having absolutely no key to connect the two. Cracking that genetic cipher became one of the greatest scientific quests of the 20th century, and the key, as it turned out, was labeled protein synthesis. It provided the direct, undeniable proof needed to finally read life's instruction manual.
This work was spearheaded by scientific giants like Marshall Nirenberg and Har Gobind Khorana. They devised brilliant experiments to assign a specific "word" from the genetic alphabet to each amino acid. We now call these three-letter genetic words codons.
The First Codon Is Cracked
The big moment came in 1961. Nirenberg and his colleague, Heinrich Matthaei, set up a cell-free system, much like the ones used in earlier labeling studies. But they added a stroke of genius: a synthetic, man-made strand of messenger RNA (mRNA) made of just one repeating letter—uracil (U).
They fed this simple "poly-U" message into the system along with a mix of all 20 amino acids. Here's the clever part: only one of those amino acids was radioactively labeled. They ran the experiment 20 times, swapping out which amino acid got the radioactive tag. When they finally used labeled phenylalanine, something amazing happened. The system churned out a long, radioactive chain made of nothing but phenylalanine.
The conclusion was instant and irrefutable. The three-letter codon "UUU" had to be the genetic instruction for the amino acid phenylalanine. The first word in the language of life had been translated.
This single experiment, made possible by labeled protein synthesis, blew the doors wide open. It created a direct, measurable link between a genetic sequence and an amino acid, giving scientists the blueprint to decipher the entire code.
This discovery kicked off a global scientific race. By 1966, the research teams of Nirenberg, Khorana, and Severo Ochoa had systematically mapped all 64 possible codons. They found that the codon "AUG" not only codes for methionine but also serves as the universal "start" signal that kicks off protein synthesis 100% of the time. Khorana's group even managed to synthesize custom messenger RNAs from scratch, proving the system worked with near-perfect accuracy. For a deeper dive into these historic experiments, the National Library of Medicine's digital archive has a fantastic historical review.
Why This Ancient Code Matters For Modern Performance
This isn't just a dusty history lesson; this genetic code is the software running your biological hardware right now. Understanding how it works is the reason we know that certain amino acids are so vital for physical performance. For example, knowing that leucine has a powerful effect on initiating muscle protein synthesis allows us to create targeted nutritional strategies for athletes and bodybuilders.
The entire world of performance peptides and proteins is built on this foundational knowledge. You can learn more about how they differ by reading our guide on the difference between peptides and proteins.
Our ability to not just read, but now write, this code is what enables us to engineer biological systems with incredible precision—from developing new medicines to designing research peptides that target specific cellular functions. Cracking the code gave us the rules of the game.
Modern Methods For Labeling Proteins
Gone are the days of researchers painstakingly synthesizing radioactive materials from scratch. Today, we have a sophisticated toolbox of modern labeling strategies, and choosing the right one is like a master mechanic picking the perfect tool for a specific engine repair. The three big players in this field are metabolic labeling, chemical labeling, and enzymatic labeling.
Each of these approaches gives us a different way to attach a molecular "tag" onto a protein. Getting a handle on how they differ is crucial for understanding how scientists can so precisely measure things like muscle growth or track a peptide's journey through a cell. The method you pick will ultimately define the specificity, simplicity, and quality of the data your experiment generates.
H3: Metabolic Labeling: Building From The Inside Out
Imagine you’re building a fleet of cars, but instead of using standard steel, you use a uniquely colored, heavier type of steel. Every car that rolls off the assembly line made from this material will be instantly identifiable. This is the core idea behind metabolic labeling.
Instead of feeding a cell its usual diet of amino acids, we provide it with a "tagged" version. These tags are usually stable isotopes—heavier, non-radioactive cousins of common atoms like carbon-13 or nitrogen-15. The cell’s machinery doesn't know the difference and happily uses these heavy amino acids as standard building blocks. As the ribosomes churn out new proteins, they naturally incorporate these labeled aminos.
This approach is incredibly powerful for seeing protein synthesis in action. It gives you a real-time snapshot of what the cell is actively producing, making it the gold standard for measuring protein synthesis rates.
The catch? This method isn't surgical. It tags all newly made proteins, which is fantastic for getting a big-picture view of cellular activity but not so great if you’re trying to zero in on just one specific protein.
H3: Chemical Labeling: Modifying The Finished Product
Now, let's picture a fully assembled car coming off the line. This time, you decide to apply a custom vinyl wrap directly onto its body. That's the essence of chemical labeling. This technique starts with a purified, fully formed protein and attaches a label directly to it using a chemical reaction.
This gives you complete control over which protein gets tagged. The most common targets for these chemical reactions are the reactive side chains on certain amino acids, like the amines on lysine or the thiols on cysteine. A great example is attaching a fluorescent dye like Cy5 to a peptide, which allows you to literally watch where it goes inside a cell.
This whole process relies on our deep understanding of how proteins are built in the first place, following the blueprint laid out by our genes.
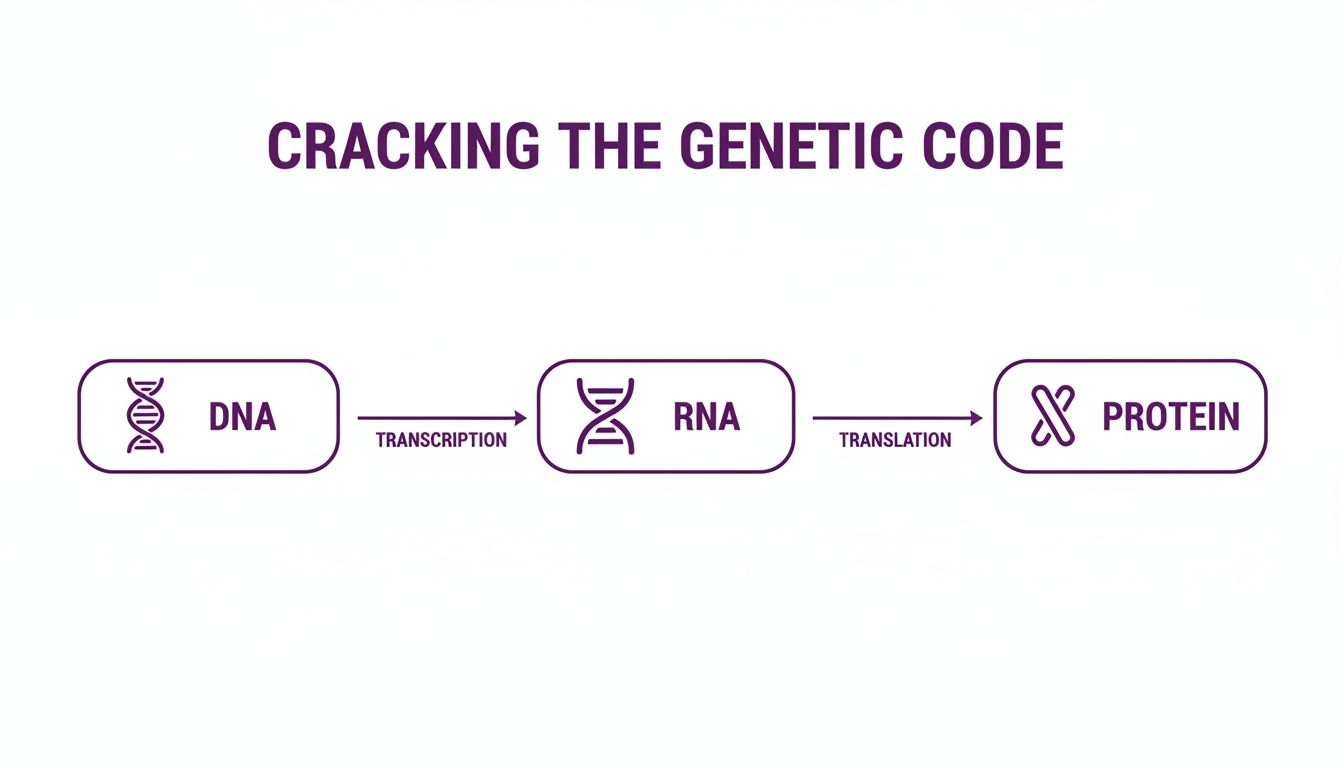
As you can see, the genetic code is first transcribed from DNA to RNA, and then that RNA is translated into the amino acid chain that becomes the final protein—the very molecule we’re looking to tag.
The huge win with chemical labeling is its specificity; you can label just one type of protein. The trade-off is that it’s a post-translational modification, meaning it happens after the protein is made. So, it can't tell you anything about the rate of synthesis, but it’s perfect for tracking, imaging, and purification.
H3: Enzymatic Labeling: The Precision Tool
Finally, imagine having a high-precision robotic arm that can place a tiny, unique badge onto one specific spot on the car—and nowhere else. This is the level of accuracy we get with enzymatic labeling. This method uses other proteins, called enzymes, as a kind of biological "super glue" to attach a label to a target protein with incredible precision.
A popular way this is done is by genetically engineering a short "tag sequence" into our protein of interest. A highly specific enzyme is then introduced that recognizes only that tag. It then attaches the label—like biotin or a fluorescent molecule—right where it’s supposed to go. This approach avoids the potentially harsh chemicals used in other methods and ensures the label is placed perfectly, minimizing any disruption to the protein's natural function. This precision is absolutely essential for complex studies where you need to be sure the protein is still behaving normally.
To help you sort through these options, here's a quick look at how the main strategies stack up against each other.
Comparison of Protein Labeling Strategies
This table breaks down the three main strategies for labeling proteins, highlighting their core mechanics, strengths, and weaknesses.
| Labeling Strategy | How It Works (Analogy) | Specificity | Best For |
|---|---|---|---|
| Metabolic Labeling | Building a car with colored parts | Low (labels all new proteins) | Measuring overall synthesis rates and protein turnover. |
| Chemical Labeling | Applying a vinyl wrap to a finished car | High (labels a specific purified protein) | Tracking protein location (imaging) and purification. |
| Enzymatic Labeling | A robot adding a badge to a precise spot | Very High (site-specific) | Live-cell imaging and when preserving protein function is critical. |
Ultimately, choosing the right method boils down to what you're trying to discover. Whether you need a sweeping overview of a cell's entire output or a highly focused look at a single molecule's behavior, there's a labeled protein synthesis technique designed for the job. Of course, great experiments start with great materials, and producing these often requires advanced peptide synthesis services.
Real-World Applications In Performance Research
So, we've covered the history and the science, but let's get to the real question: what does this actually mean for metabolic health and physical performance? This is where the rubber meets the road—where labeled protein synthesis stops being a textbook concept and becomes the tool that verifies if a compound actually works.
From mapping every single protein in a muscle cell to proving how a research peptide triggers growth, these techniques give us the definitive answers. This isn't just theory. It's about generating the cold, hard data that separates a promising idea from a proven biological effect.
Charting The Entire Proteome
One of the most mind-blowing applications is in the field of proteomics. Think of it like this: you're creating a complete, highly detailed census of every protein inside a cell or tissue at a single moment in time. By using stable isotope labels, researchers get a dynamic snapshot of this "proteome" and see precisely how it responds to something like exercise or a new compound.
For instance, a scientist could take a tiny muscle sample before and after a tough workout. Using metabolic labeling, they can measure exactly which proteins are being built at a faster rate after the training. This reveals the specific molecular machinery firing up to repair and grow the muscle, giving us a roadmap to develop even better interventions.
This is how we graduated from simply knowing that lifting weights builds muscle to understanding the intricate pathways—like mTOR activation—that truly drive hypertrophy. This depth of knowledge is exactly why we can analyze and discuss specific compounds in our guide on the best peptides for muscle growth.
Validating Peptide Mechanisms
For anyone involved in performance research, this is arguably the most crucial application. When a new research peptide hits the scene with big claims, how do we know it’s the real deal? The answer, nearly every time, involves studies using labeled protein synthesis.
Let's play out a common scenario. A new growth hormone secretagogue is supposed to build muscle. To prove it, researchers can treat muscle cells in a dish with this peptide while feeding them labeled amino acids. If the compound works, mass spectrometry will show a huge spike in the incorporation of those labeled aminos into new proteins compared to the untreated cells.
This provides clear, quantitative proof of the peptide's anabolic effect at a cellular level. It’s the difference between a hypothesis and a confirmed mechanism of action, turning a promising compound into a scientifically validated tool for investigation.
The same idea works for metabolic research. Scientists use labeled glucose and fatty acids to trace how compounds like GLP-1 agonists affect fuel choices in different tissues, painting a clear picture of their impact on body composition and energy.
Bio-Imaging And In Vivo Tracking
Then there's the really cool part: watching all of this happen live. By attaching a fluorescent tag, like the popular Cy5 dye, to a peptide, researchers can use advanced microscopes to track its journey through a living system in real-time.
You can get answers to critical questions:
- Receptor Binding: Does the peptide actually dock onto its target on the cell surface? You can see it light up.
- Cellular Uptake: Does the peptide get pulled inside the cell, or does it do its work from the outside? You can watch it happen.
- Biodistribution: In animal models, where does the peptide end up in the body? This is vital for understanding its effects and how it’s eventually cleared.
For example, by chemically attaching Cy5 to a peptide, you can literally watch it being absorbed by living cells. The dye emits light around 670 nm, a wavelength that cuts through the natural background noise of the cell, giving you a crystal-clear signal. It’s not a static image; it's a movie of molecular biology in action.
These aren't just obscure academic exercises. They are the essential checkpoints that underpin all legitimate performance research, giving us the concrete evidence needed to push our understanding of human biology forward.
Sourcing And Handling Research Peptides Safely
When your research hinges on labeled protein synthesis, the quality of your starting materials isn't just a detail—it's everything. Your entire experiment is designed to detect incredibly subtle changes inside a cell. Any contaminant or error in your peptides can throw a wrench in the works, leading to data that's not just noisy, but completely wrong.
That’s why sourcing and handling your peptides correctly is a non-negotiable part of the job. You have to demand uncompromising quality from the start.
The gold standard for any serious lab work is a purity level exceeding 99%, confirmed by independent testing. Think about it: if your peptide is only 95% pure, that other 5% is a black box of unknowns. Best case, it's harmless. Worst case, it's a rogue sequence that binds to your target, blocks your real peptide, or otherwise torpedoes your experiment from the inside.
A reputable supplier will always provide a detailed Certificate of Analysis (CoA) for every batch. This isn't just paperwork. It's your guarantee, detailing the exact purity, mass, and sequence as verified by analytics like HPLC and mass spectrometry. Never work without it.
Understanding "Research Use Only" (RUO)
As you navigate the world of lab reagents, you'll constantly encounter the "Research Use Only" (RUO) designation. This isn't a suggestion; it's a strict mandate. RUO compounds are intended exclusively for in-vitro laboratory experiments. Period. They are absolutely not for human or veterinary use.
This distinction is critical for a few key reasons:
- Safety: RUO compounds are not drugs. They haven't gone through the exhaustive clinical trials needed to prove they are safe or effective for any living being.
- Legality: Using or distributing these compounds for anything other than their intended research purpose can carry serious legal consequences.
- Scientific Integrity: The RUO system ensures these potent tools are used responsibly in controlled environments to advance our collective knowledge, not for dangerous and unproven self-experimentation.
These are not supplements or "bio-hacks." They are powerful chemical tools designed for a specific job, like watching labeled protein synthesis happen in a petri dish.
Proper Storage and Handling
Once you've sourced a high-purity RUO peptide, the responsibility for its integrity falls on you. Peptides are delicate molecules. Think of them like a high-performance engine; you wouldn't just leave it out in the rain and expect it to work flawlessly. The same principle applies here.
Mishandling can degrade your peptides, turning your expensive and carefully chosen materials into worthless powder.
Follow these cardinal rules for peptide handling:
- Storage: Most peptides arrive as a lyophilized (freeze-dried) powder. In this state, they're quite stable, but they should still live in a freezer at -20°C or colder. This is your best defense against slow degradation over time.
- Reconstitution: When you're ready for an experiment, you'll reconstitute the powder in a sterile, appropriate solvent. Be warned: once it's a liquid, the peptide is far more fragile and vulnerable to bacterial contamination or damage from repeated freeze-thaw cycles.
- Aliquoting: To protect your investment, never thaw and re-freeze your entire stock. The best practice is to reconstitute the whole vial, then immediately divide it into smaller, single-use aliquots. This way, you only thaw what you need for one experiment, keeping the rest of your supply pristine and safe in the freezer.
By insisting on verified purity from your supplier and sticking to these strict handling protocols in your lab, you build your experiments on a foundation of reliability. This commitment is what separates clean, trustworthy data from a failed project.
Common Questions About Labeled Protein Synthesis
As you get deeper into performance research, the concept of labeled protein synthesis inevitably comes up. It can sound pretty technical, but once you get past the jargon, the core ideas are surprisingly intuitive. Let's walk through some of the most common questions to clear things up.
What Is The Simplest Way To Understand This Process?
The best way to think about it is like putting a tiny GPS tracker on your cellular building blocks. Scientists attach a special "tag" to amino acids, which are the raw materials cells use to construct every protein in the body.
When a cell gets the signal to build new proteins—say, for repairing muscle tissue or regulating metabolism—it pulls from the available pool of amino acids. By using these pre-tagged aminos, researchers can then follow the signal to see exactly where, when, and how fast new proteins are being assembled. It's the most direct way to get definitive proof that a process like muscle growth is actually happening.
Why Are There Different Types Of Labels?
You need different tools for different jobs, and the same is true for protein labels. The specific tag a scientist chooses depends entirely on what they’re trying to find out.
- Isotopic Labels: These are the gold standard for quantitative work. They use heavier, stable atoms (like Carbon-13) that don't change the amino acid's function. Using a highly sensitive instrument called a mass spectrometer, a researcher can measure exactly how much new protein was created.
- Fluorescent Labels: Think of these as high-visibility vests for your proteins. Dyes like Cy5 literally light up under a microscope, which is perfect for visualizing where a protein is located inside a cell or watching it move around in real time. Its red to near-infrared glow (around 670 nm) is fantastic because it stands out against the cell's natural background fluorescence.
How Does This Relate To Research Peptides?
This is where it all comes together. Labeled protein synthesis is the scientific method used to prove that a research peptide actually does what it's claimed to do. It’s the bridge between a hypothesis and hard evidence.
For instance, to confirm a growth hormone secretagogue truly triggers protein synthesis, a researcher will treat a cell culture with the peptide in a medium containing labeled amino acids. By measuring how many of those labeled aminos get incorporated into new proteins, they can definitively prove the peptide's mechanism of action. This is precisely why starting with high-purity peptides is non-negotiable for getting clean, reliable data.
Is Using Labeled Compounds Safe in Research?
In a controlled lab setting, yes, it's completely safe. These experiments are conducted by highly trained professionals who adhere to strict safety protocols for handling all chemical reagents.
The vast majority of modern labeled protein synthesis studies use stable (non-radioactive) isotopes, which are perfectly harmless and pose no radiation risk. In the specific cases where radioactive isotopes are necessary, their use is heavily regulated and monitored to ensure the safety of everyone involved. It's crucial to remember these compounds are tools for lab work (in vitro) and are never meant for human use, as the "Research Use Only" label makes crystal clear.
For researchers who demand uncompromising purity and reliability in their experiments, Bullit Peptides offers third-party verified compounds designed for precision. Make sure your results are built on a foundation of quality you can trust.
Explore our catalog of research-grade peptides at https://bullitpeptides.com.







