When you're working with a research compound as potent as retatrutide, the dose you choose isn't just a number—it's the most critical variable in your entire experimental setup. Clinical trials have zeroed in on weekly maintenance doses of 4 mg, 8 mg, and 12 mg, and the data couldn't be clearer: higher doses lead to more profound metabolic effects.
Understanding the Retatrutide Dose for Research
For any scientist, thinking about retatrutide dosage isn't about finding a therapeutic sweet spot. It's about calibrating a powerful scientific instrument. As a novel triple-agonist peptide hitting GIP, GLP-1, and glucagon receptors all at once, its effects are intensely dependent on the concentration you use.
Think of it less like mixing a simple chemical solution and more like adjusting the faders on a complex audio mixer. Each receptor—GIP, GLP-1, and glucagon—has a different sensitivity and response as the dose climbs. This dose-response relationship is precisely what allows researchers to isolate and probe specific metabolic pathways.
The Dose-Dependent Effect in Action
The jaw-dropping data that launched retatrutide into the spotlight came directly from its Phase 2 trial. In June 2023, the results shared at the American Diabetes Association's 83rd Scientific Sessions revealed a direct and powerful link between dose and weight loss.
In that landmark study, participants received weekly subcutaneous injections, starting low and slowly ramping up to maintenance doses of 4 mg, 8 mg, or 12 mg over 48 weeks. The results were stunning. The group on the highest 12 mg dose achieved an average body weight reduction of 24.2%. For someone starting at 240 pounds, that’s a loss of over 58 pounds.
This table breaks down the key findings from that trial, showing just how closely weight loss was tied to the dose administered.
Retatrutide Phase 2 Trial Dosing and Key Outcomes
| Dose Cohort (Weekly) | Mean Body Weight Reduction at 48 Weeks | Equivalent Weight Loss Example (240 lb individual) |
|---|---|---|
| 4 mg | 17.5% | ~42 lbs |
| 8 mg | 22.8% | ~55 lbs |
| 12 mg | 24.2% | ~58 lbs |
The data paints a very clear picture. While even the lower doses produced significant results, the escalating effect as the dose increased from 4 mg to 12 mg was undeniable. You can dive deeper into the clinical trial data in this comprehensive retatrutide peptide guide from Sky Health.
A higher dose doesn't just turn up the volume on the same signal; it fundamentally changes the metabolic conversation. At lower concentrations, you're primarily seeing the effects of GIP and GLP-1 agonism. But as you push into higher doses, the glucagon receptor’s influence on energy expenditure really comes alive, offering a completely unique mechanism to study.
This visualization from the study drives the point home, showing the clear separation in efficacy between the different dose cohorts.
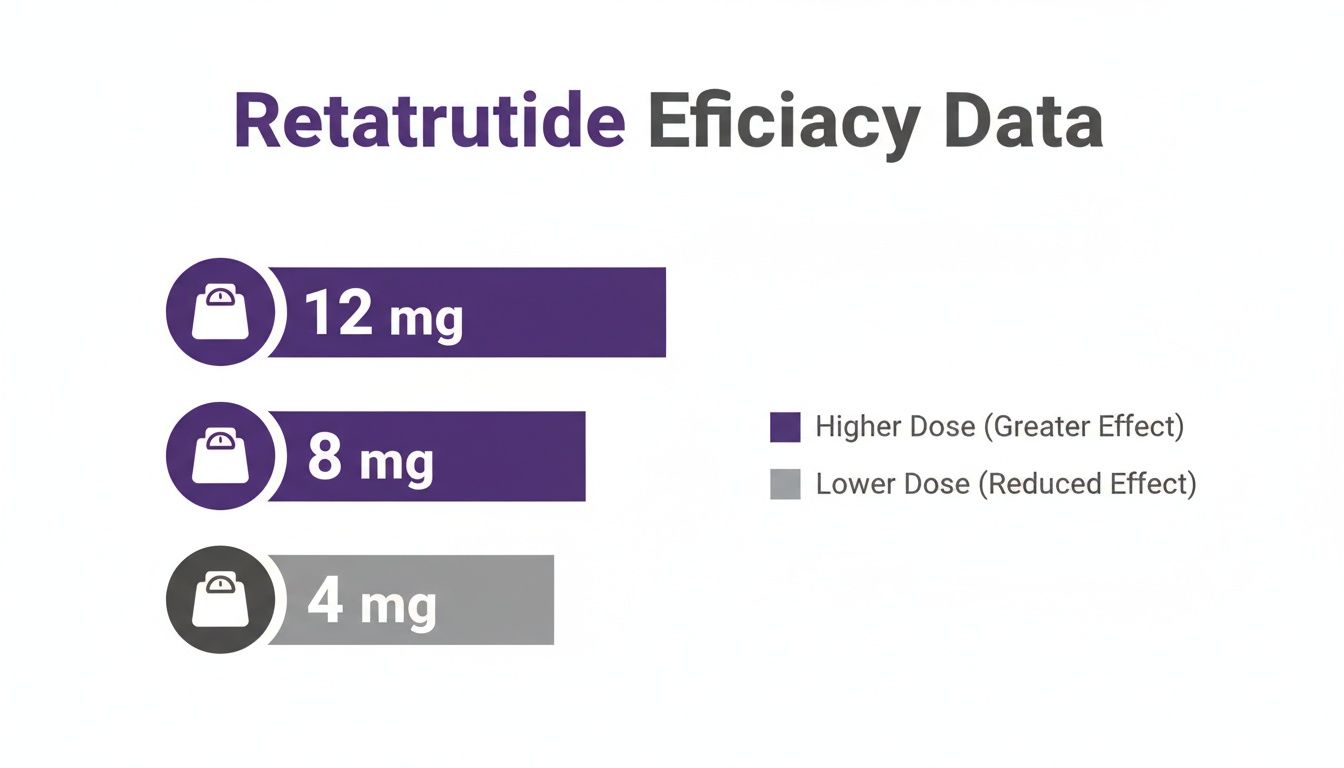
The graph makes it obvious that while the 4 mg dose is certainly effective, the 8 mg and 12 mg doses are in another league entirely. This is why for any scientist, establishing the proper 'Research Use Only' context is so vital. The dose is the key that unlocks different biological doors, from subtle changes in cellular signaling to massive, systemic shifts in metabolism.
The Science Behind Triple Agonism and Dose Response
The impressive results we're seeing with retatrutide aren't happening by accident. They're a direct result of its incredibly smart triple-agonist design. Unlike older compounds that hit just one metabolic target, retatrutide activates three separate receptors: GLP-1, GIP, and Glucagon. This strategy creates a powerful, combined effect that gets stronger as the dose goes up.
Think of it like a sound engineer mixing a track. Each receptor is a different channel on the mixing board. The retatrutide dose is the set of faders, allowing a researcher to dial in a precise metabolic "sound"—influencing everything from hunger signals to how many calories the body burns.
The Three Pillars of Retatrutide's Power
To really get a handle on the dose-response relationship, you have to appreciate what each of these three receptors brings to the table. Each one solves a unique piece of the metabolic puzzle. It’s their combined activation that makes this compound so potent.
GLP-1 (Glucagon-Like Peptide-1): This is the foundation, the deep bass of satiety. GLP-1 agonists have a long track record for slowing down digestion and sending powerful "I'm full" signals to the brain, which helps curb appetite and cravings.
GIP (Glucose-Dependent Insulinotropic Polypeptide): This is the mid-range, fine-tuning how the body handles nutrients and insulin. GIP helps regulate the processing of fats and sugars, contributing to better overall metabolic function.
Glucagon: This is the high-frequency treble, the special sauce that truly makes retatrutide different. Activating the glucagon receptor directly ramps up energy expenditure and thermogenesis. It basically turns up the body's metabolic thermostat.
At a lower retatrutide dose, the effects are mostly coming from GLP-1 and GIP working together, with the focus on appetite suppression and insulin sensitivity. But as you climb the dosage ladder, that glucagon "treble" really starts to sing. This is a critical distinction for any research.
The glucagon activation is the key differentiator. At higher doses, this mechanism drives a significant increase in energy expenditure that isn't seen with single or dual agonists. This explains the greater weight loss observed at the 8 mg and 12 mg levels in clinical trials.
How Higher Doses Unlock Glucagon's Full Potential
The real story behind the escalating retatrutide dose is all about engaging that glucagon receptor. While the molecule has an affinity for all three targets, its effect on glucagon becomes especially powerful at higher concentrations. This is a crucial piece of information when designing a study.
For example, a scientist looking into the cellular mechanics of fat burning (lipolysis) would need to use a dose high enough to fully switch on this pathway. An experiment run with a low dose might miss this key effect entirely, leading to incomplete or misleading conclusions.
A higher retatrutide dose doesn't just turn up the volume on GLP-1 and GIP; it brings a potent new player into the game by strongly activating the glucagon pathway. This is precisely why the 12 mg dose achieved a staggering mean weight reduction of 24.2% in trials—it had all three metabolic engines roaring at full speed.
This is fundamental knowledge for any researcher trying to isolate specific metabolic effects or replicate the profound systemic outcomes seen in the clinical literature. Choosing the right dose isn't just about picking a number; it’s about strategically targeting the right biological mechanism for your research question.
For those exploring other compounds in this space, our guide on the best peptides for weight loss research can provide more context on how these different mechanisms stack up. Ultimately, the precision offered by the retatrutide dose gives researchers a level of experimental control that is essential for generating clear, reliable, and reproducible data.
Mastering Dose Titration in Research Protocols
When you're working with a powerful compound like retatrutide in a high-stakes research setting, you can't just flip a switch and expect perfect results. The introduction has to be handled with care and precision. That's where dose titration becomes the absolute cornerstone of a sound experimental protocol, adapting what we’ve learned from clinical trials for reliable lab work.
Dose titration is simply the practice of starting with a very low dose and methodically increasing it over weeks or even months. Think of it like breaking in a high-performance engine. You don't redline it straight out of the garage; you let the system get used to the increasing demands to ensure it runs smoothly and avoids damage. The scientific principle is the same: you give the biological system time to adapt, which minimizes adverse effects and, crucially, protects the integrity of your data.
This step-by-step increase isn't just a friendly suggestion—it's a proven blueprint straight from successful clinical trials. This methodical approach is what rigorous science is built on.

Why Gradual Escalation is Non-Negotiable
The main reason we titrate is to get a handle on the gastrointestinal (GI) side effects that are so common with GLP-1 receptor agonists. Hitting the system with a high dose right away can overwhelm it, leading to nausea or vomiting. This is a huge problem in a study because it introduces confounding variables—is the subject eating less due to the peptide's intended anorectic effect, or simply because it feels sick?
By starting low and inching the dose up, the system builds tolerance. This ensures the metabolic changes you're observing are a direct result of retatrutide’s unique triple-agonist mechanism, not just a secondary reaction to physiological distress. This is how investigators can reach effective target concentrations while keeping their animal models healthy and isolating the specific effects of the compound.
A Blueprint for Titration from Clinical Trials
The Phase 2 trial for retatrutide gives us a fantastic, field-tested titration schedule that can be easily adapted for preclinical research models. The entire protocol was designed to safely guide subjects toward higher, more effective maintenance doses.
Here’s what a typical escalation schedule looks like, based on that clinical data:
- Initiation Phase (Weeks 1-4): Start with a 2 mg weekly dose. This is the introductory, "get-acquainted" period.
- First Escalation (Weeks 5-8): Double the dose to 4 mg weekly.
- Second Escalation (Weeks 9-12): Depending on your target, increase to either 6 mg or 8 mg weekly.
- Final Escalation (Weeks 13+): Move to the final maintenance dose you're aiming for, such as 8 mg, 10 mg, or 12 mg.
This schedule, with its deliberate four-week intervals, provides a reliable roadmap. For researchers, getting these small increments exactly right is mission-critical, and that's where specialized tools come into play. To ensure this level of precision, you can explore our guide on using a peptide calculator app for research.
The goal of titration isn't to completely eliminate side effects, but to manage them so effectively that they don't compromise the research. A slow, steady ramp-up is the key to collecting clean, interpretable data at the higher doses where the real magic happens.
The logic here isn't new; it mirrors the protocols for other leading GLP-1 agonists. Their dosing schedules also rely on once-weekly injections with a gradual titration over 4 to 12 weeks to keep GI issues in check. Nausea might be high at first, but it almost always subsides over time with this approach.
By following a structured titration protocol, a researcher can confidently study the effects of a specific retatrutide dose, knowing the biological system was properly prepared for it. This methodical escalation is what separates haphazard experimentation from disciplined scientific inquiry.
Analyzing Dose Dependent Safety and Tolerability
Any powerful tool demands a healthy respect for its limits. When it comes to a research compound like retatrutide, that means getting a crystal-clear picture of its safety and tolerability profile. In any well-designed experiment, a researcher has to anticipate and account for potential side effects—they're unwanted variables that can muddy the waters and skew your data.
Fortunately, the clinical trials have given us a pretty transparent look at how different retatrutide doses correlate with specific adverse events. This isn't about giving medical advice. It’s about arming researchers with the data they need to design smarter, more robust experiments. By understanding the direct line between dose, titration speed, and the frequency of side effects, you can better control the variables in your own in-vitro or preclinical work.
The main culprits here are gastrointestinal issues, which is a classic calling card for this entire family of incretin mimetics.

The Gastrointestinal Effect and Dose Correlation
The most common side effects popping up in the retatrutide trials were nausea, diarrhea, and vomiting. What’s crucial to understand is that the incidence and severity of these GI effects are directly tied to two things: the maintenance dose you’re aiming for and the speed of the titration schedule you use to get there.
Push the dose higher and ramp up faster, and you're almost guaranteed to see a higher rate of these initial side effects.
This dose-dependent pattern is a critical piece of the puzzle for any investigator. Imagine a study subject experiences significant nausea and, as a result, naturally eats less. A researcher might mistakenly chalk up the subsequent weight loss entirely to retatrutide's known appetite-suppressing effects, when in reality, the temporary side effect was a major confounder.
Knowing this ahead of time lets you design protocols that minimize these issues, ensuring the data you collect is as clean and accurate as it can possibly be. It highlights why a slow, methodical titration isn't just a friendly suggestion—it's a cornerstone of rigorous scientific work.
Side Effect Profile by Retatrutide Dose
The published trial data gives us a clear, numbers-based breakdown of how these GI issues climb with the dosage. While even the placebo groups reported some level of discomfort (which is common in trials), the trend across the retatrutide arms is impossible to miss.
Here's a look at how the most common gastrointestinal side effects stacked up at different maintenance doses.
| Adverse Event | Incidence in Placebo Group | Incidence in 4 mg Group | Incidence in 8 mg Group | Incidence in 12 mg Group |
|---|---|---|---|---|
| Nausea | 9.9% | 35.7% | 47.3% | 58.9% |
| Diarrhea | 8.5% | 18.6% | 23.3% | 21.9% |
| Vomiting | 2.8% | 11.4% | 16.4% | 26.0% |
As you can see, the data tells a compelling story. A subject on the 12 mg retatrutide dose is nearly six times more likely to experience nausea than someone in the placebo group. That's a massive variable that absolutely has to be factored into any research design.
The key takeaway is that these side effects are typically transient. Most GI issues were reported as mild-to-moderate and flared up primarily during the initial dose-escalation phase. Once a subject's system adjusted to their maintenance dose, the incidence of these events dropped off significantly.
This pattern reinforces the idea that the body can and does adapt to the compound. A slow titration schedule simply gives it the time it needs to do so, minimizing disruptive side effects and allowing the true metabolic impact of the peptide to be observed without interference.
The Role of Purity in Reliable Outcomes
This whole discussion on side effects brings us to another mission-critical point for researchers: the absolute necessity of using high-purity compounds. When you're trying to isolate the effects of a specific retatrutide dose, the last thing you want are confounding variables thrown into the mix by contaminants in your sample.
Sourcing peptides with a verified purity of >99% is non-negotiable for serious research. An impure product could introduce unknown substances into your experiment, which might cause their own side effects or, even worse, alter the pharmacological action of the retatrutide itself. This would make it impossible to know if your results were due to the peptide, the contaminants, or some unpredictable synergy between them.
For any serious scientific work, ensuring the purity of your materials is just as fundamental as perfecting your titration protocol. It’s a foundational step toward generating valid, reproducible, and ultimately publishable data.
Designing Your Research Use Only (RUO) Experiments
Knowing the clinical trial data for retatrutide is the first step. But the real challenge? Translating that information into a solid lab experiment that actually works. The dose-response curve we’ve seen in human trials isn't just a piece of trivia—it's your playbook for any serious scientific investigation. It’s what helps you bridge the gap from theory to practice, letting you design powerful in-vitro and preclinical studies that produce clean, repeatable results.
The trick is to let the clinical dosing tiers inform your lab concentrations. This is how you tether your experiment to established human data, which instantly makes your findings more relevant. Whether you're working with cells in a dish or with animal models, the fundamental principle holds true: the dose dictates the effect.
Applying Dose Concepts to In-Vitro Studies
Let's get practical. Imagine you're a researcher trying to figure out how retatrutide encourages "adipocyte browning"—the process of converting lazy, energy-storing white fat into active, energy-burning brown fat. This is a classic in-vitro experiment, perfect for a cell culture setup.
Instead of just picking a random concentration and hoping for the best, you could design the experiment to mirror the tiered glucagon activation seen in the human trials.
- Low-Dose Simulation: One set of cells gets a retatrutide concentration that mimics the effects of a 4 mg human dose. At this level, you’re mostly hitting the GIP and GLP-1 pathways.
- High-Dose Simulation: Another set of cells is exposed to a much higher concentration, simulating the potent 12 mg dose. Here, you expect to fully engage the glucagon receptor and kick thermogenesis into high gear.
By comparing how the cells in each group respond, you can directly see how that extra glucagon activity at higher doses flips the switch on specific genes and mitochondrial functions tied to fat browning. This isn't just a guess; it's a method for gathering concrete, dose-dependent proof of how the compound really works.
Preclinical Models and Dose-Dependent Outcomes
This same logic scales up perfectly for in-vivo work with animal models, like mice or rats. Let's say a team wants to study retatrutide's effect on non-alcoholic fatty liver disease (NAFLD). They could design their study to reflect the clinical dosing tiers. After putting all the mice on a high-fat diet to induce fatty liver, one group would get a lower weekly dose of retatrutide, and another would get a significantly higher one.
From there, they could track key biomarkers like liver enzymes and the actual amount of fat in the liver. This setup allows them to draw a straight line between the dose of retatrutide and the reduction in liver fat, offering clear evidence of its potential. For young men in weight training, for example, this glucagon-driven effect at higher doses is thought to crank up energy expenditure by 20-25%. It achieves this by slashing liver fat (with reductions up to 82% at the 12 mg dose), which helps preserve lean muscle far better than single-agonist peptides can. As suppliers now offer research-grade retatrutide with >99% purity, labs can finally dig into the mechanisms behind these dose-specific effects, from delayed gastric emptying to glucagon's role as the 'gas pedal' for thermogenesis. You can get a deeper look into retatrutide’s unique mechanisms and how they work together.
The integrity of your experiment hinges entirely on the quality and accuracy of the materials you use. An inaccurately dosed or impure peptide will introduce so many confounding variables that your results will be rendered meaningless. Precision isn't just a goal; it's a prerequisite for valid science.
This is exactly why sourcing high-purity, accurately lyophilized peptide is non-negotiable. When your entire research project depends on observing subtle, dose-dependent changes, you have to be absolutely certain that the dose you think you're giving is the dose the biological system is actually getting. Any slip-up here doesn't just skew the data—it invalidates the entire experiment.
Best Practices for Sourcing and Handling Research Peptides
Knowing the right retatrutide dose is one thing, but if you can’t trust the material in your vial, all that theory goes out the window. The practical side of handling these compounds is where your research truly succeeds or fails. After all, your experimental data is only as good as the peptide you start with.
Sourcing high-purity, accurately dosed retatrutide is non-negotiable. Think of it like cooking a gourmet meal—you wouldn't use subpar ingredients and expect a five-star result. For legitimate research, you need peptides with a verified purity of over 99%. Anything less introduces confounding variables that could completely skew your findings, leaving you to wonder if your results came from the peptide or its impurities.
Ensuring Peptide Integrity and Stability
Once you have a high-quality peptide in hand, the real work begins. These molecules are incredibly sensitive. Mishandle them, and you can quickly degrade the compound, making your carefully planned retatrutide dose worthless before you even begin.
To protect the peptide's structural integrity, you have to follow a strict protocol:
Lyophilized for a Reason: Your peptide will arrive as a freeze-dried (lyophilized) powder. This isn't just for show; it's a critical step that keeps the molecule stable and safe during transit.
Mind the Temperature: Before it's mixed, the vial belongs in a freezer (around -20°C or -4°F). Once you reconstitute it into a liquid, it moves to the refrigerator (between 2-8°C or 36-46°F) and has a limited shelf life.
Gentle Reconstitution: This is where you add a sterile solvent, usually bacteriostatic water, to the powder. The key word here is gently. Shaking the vial can shear the delicate peptide chains. For a step-by-step guide, check out our tutorial on how to reconstitute peptides correctly.
Precision is everything. A tiny slip-up in your math or a clumsy reconstitution can completely alter the solution's concentration. This makes accurate dosing impossible and guarantees inconsistent, unreliable data.
From Powder to Precise Concentration
The final step is creating a solution with the exact concentration your experiment calls for. This requires meticulous calculation, factoring in the total amount of peptide in the vial and the precise volume of solvent you introduce.
Every single action—from checking the Certificate of Analysis (COA) for purity to the final, gentle swirl of the vial—is about controlling variables. When you master these handling practices, you can be confident that the retatrutide dose you planned is the exact dose your experiment gets. That’s how you set the stage for clear, defensible scientific results.
Frequently Asked Questions on Retatrutide Dosing
When you're working with a research compound as complex as retatrutide, questions are bound to pop up. Let's tackle some of the most common ones that arise in a lab setting, keeping everything strictly within the context of "Research Use Only" to guide scientific work.
What Is the Standard Administration Frequency in Studies?
Look across the board—from the foundational preclinical work to the major human clinical trials—and you'll see a consistent pattern: retatrutide is administered once per week. This isn't just a matter of convenience. It's a schedule dictated by the compound's remarkably long biological half-life.
That staying power means it can maintain steady, effective plasma concentrations with a simple weekly administration. For researchers working with animal models, mirroring this weekly protocol is key to accurately recreating the peptide's pharmacokinetics and tracking its cumulative metabolic impact over the long haul.
Why Is Slow Dose Escalation So Critical for Research?
In any good study, you have to control your variables. That's precisely why a slow dose escalation, or titration, is non-negotiable when working with retatrutide. If you introduce a powerful triple-agonist at a high dose right out of the gate, you risk triggering significant GI side effects in your test subjects.
Those side effects become confounding variables that can muddy your data. For instance, is a subject eating less because of the peptide's intended metabolic action, or simply due to nausea? A gradual, stepped increase allows the biological system to acclimate, stripping away those adverse effects so you can be sure your observed outcomes are a direct result of the specific retatrutide dose you're testing.
The whole point of titration in a research setting is to isolate the variable you're testing. It's how you ensure that the metabolic changes you measure are from retatrutide's actual mechanism, not just a side effect of physiological stress from hitting the system too hard, too fast.
How Does Glucagon Agonism Affect Dose Selection?
The glucagon receptor agonism is a huge part of what makes retatrutide so unique, and it’s a major factor in designing a dosing protocol. While you’ll see the effects of its GLP-1 and GIP activity at lower concentrations, the powerful thermogenic effects driven by glucagon agonism really only kick in at the higher end of the dosing spectrum.
So, if your research goal is to investigate mechanisms like liver fat oxidation or a significant boost in energy expenditure, your experimental design has to use concentrations from the upper tiers of the clinical trial data. Sticking to a lower retatrutide dose would mean that this powerful pathway is never fully engaged, which could lead you to incomplete or even misleading conclusions about what the compound is truly capable of.
For any researcher whose work depends on accurate, repeatable results, the quality of your materials is the bedrock of your experiment. Bullit Peptides offers research-grade retatrutide that is third-party tested and verified to exceed 99% purity, giving you the confidence that your study is built on a foundation of absolute precision. You can find their lab-grade materials here.







